|
Acacia longifolia

Citation:
C. L. Willdenow, Sp.Pl. 4:1052 (1806). Mimosa longifolia Andr., Bot. Rep. t.207 (1802).
Derivation: longus (Latin)--long; folium (Latin)--a leaf. Synonymy: Racosperma longifolium (Andr.)Martius, Hort. Reg. Monac. Semin. (1835); R. sophorae (Labill.) Martius, Hort. Reg. Monac. Semin. (1835). Common name: Sallow wattle, Sydney golden wattle
Description:
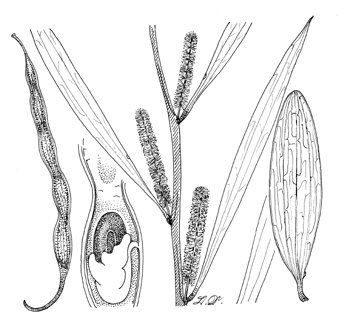
Tall erect, or low bushy, spreading shrubs, 3-7 m high; branchlets angular, glabrous, or minutely pubescent when young; bark dark grey, roughened. Phyllodes linear-lanceolate, oblong, narrowly elliptic, or obovate-oblong, 6-15 cm long, 8-35 mm broad, straight or almost so, 2-3 prominent, longitudinal parallel veins with many fine anastomosing veins between the main ones, apex acuminate or obtuse, usually mucronate; glands small but conspicuous on the upper margin near base. Inflorescences simple, 1-2 per axil, sub-sessile; spikes rather dense or sometimes loose and interrupted, bright yellow, 2-4 cm long; receptacles glabrous; flowers 4-merous. Legumes 5-10 cm long, 4-6 mm broad, cylindrical, straight or slightly curved, beaked, striate, brown, margins constricted between seeds. Seeds longitudinal in legume, obloid-elliptic; funicle short much folded, thickened into a large turbinate aril which is nearly as large as the seed.
|
|
Distribution:
|
Chiefly restricted to coastal sand dunes. Eyre Peninsula, Yorke Peninsula, Kangaroo Island, Southern Lofty, South-Eastern regions. Also Qld, N.S.W., Vic. and Tas.
Adventive in South Australia, occurring in the Eyre Peninsula (southern part), Northern Lofty, Southern Lofty, Kangaroo Island and South-Eastern regions. Also N.S.W. and Vic.
Var. sophorae (Labill.) F. Muell., Pl. Indig. Col. Vic. 2:30 (1863). Mimosa sophorae Labill., Pl. Nov. Holl. 2:87. t.237 (1806). Coastal wattle.
Low bushy spreading shrubs; phyllodes obovate oblong or oblong-elliptic, 5-10 cm long, 12-35 mm broad, much more broadly obtuse than var. longifolia.
Murray et al. (1978) presented evidence supporting the maintenance of A. sophorae and A. longifolia as separate species rather than considering A. sophorae as a variety of A. longifolia. The study included material of A. sophorae from Piccaninnie Ponds. A total of only seven samples of the two species was tested and although some differences were demonstrated this author does not consider that variability within either species was explored sufficiently to demonstrate a convincing separation of the two. Leaf flavonoid patterns were certainly different, the variation of the mature legumes seemed no greater than that accepted within say A. melanoxylon. The slight differences in seed size, seed number and water content were hardly convincing. No differences were apparent in either the flavonoid or non-protein amino acid composition of the seeds of both acacias.
The histogenesis and morphology of the phyllode of A. longifolia was studied in detail by Boke (1940). The finer details are not relevant here. Of interest was his conclusion that the vascular differentiation of the phyllode was characteristic of that for floral apices.
The early development of the floral apex of A. longifolia was published by Newman (1936). The author interprets the legume as a single laminar structure which by incurving and adpression of its margins encloses a single cavity, the ovules arising on the incurved margins of this laminar structure.
Many legumes, but not all, are able to use atmospheric nitrogen through the mediation of bacteria growing in root nodules. Acacia does bear nodules and is able to fix atmospheric nitrogen. Lawrie (1981) reported on five Acacia species, all of which occur in South Australia, that nodular activity reached a maximum in spring and declined to a minimum in late summer. There was considerable variation both between species and individual plants.
Nitrogen fixation by Acacia used for sand dune rehabilitation was studied by Barnet et al. (1985). The species included A. suaveolens and A. longifolia var. sophorae. The sites consisted of one in which natural regrowth had occurred but included planted Acacia. The second was grossly disturbed and included planted Acacia only. Nitrogen fixation occurred at both sites within rates previously reported. Distinct seasonal changes were apparent with higher activity in the cooler months. Fast and slow growing strains of bacteria were obtained and were classed as Rhizobium and Bradyrhizobium respectively and both were obtained from each species of Acacia.
For a note on the effect of seven pre-sowing treatments on germination (as A. sophorae) see Aveyard (1968) and for seed chipping and hot water treatments see Clemens et al. (1977).
Everist (1981) recorded stock poisoning by A. longifolia believing the toxic substance to be cyanides in the leaves.
Bates (1981) recorded two collections of the putative hybrid A. longifolia x A. oxycedrus both collected in our South-East region. Similar hybrids have been recorded from Victoria.
A. longifolia is now well established in South Africa especially the Cape Province where it is aggressively invading and displacing indigenous flora. It is also occasionally established in California. A. longifolia is now naturalised in New Zealand, Webb et al. (1988).
A. sophorae is reported by Isaacs (1987) to be used by the Aborigines as a source of seeds, often used green.
|
Flowering time: July — October. |
SA Distribution Map based
on current data relating to
specimens held in the
State Herbarium of South Australia
|
Biology:
No text
Taxonomic notes:
Two varieties are recognised.Var. longifolia: Tall shrubs; phyllodes linear-lanceolate or narrowly elliptic, 6-15 cm long, 3-15 mm broad, apex acuminate.
Cultivation:
An attractive ornamental species with dark green foliage and bright yellow flowers; does best in the higher rainfall areas. The var. sophorae is recommended for planting as a low windbreak and sand-binder in coastal areas. Both have a fast growth rate.
Author:
Not yet available
Source:
|

